| g e n u i n e i d e a s | ||||||
 |
 |
 |
 |
 |
 |
 |
| home | art and science |
writings | biography | food | inventions | search |
| carry on | ||||||||||
|
Oct 2013 They say you can't get something for nothing. And perpetual motion is an idle quest. But occasionally it seems as if cooking draws energy out of thin air. Roast a beef loin to 135F, remove it from the oven, and it will grow hotter all on its own! No source of outside energy. Nothing up its sleeves. Of course, this miracle is nothing more than a mirage. A bit of stagecraft and misdirection. When you claim meat got "hotter", you meant a thermometer in the center rose from 135F to 140F. That's the magic. The misdirection was not measuring the temperature near the loin's surface, which might be steaming away at 195F. This hot surface layer was the hidden source of extra energy, and all that happened was the surface cooled off by warming the interior. By not measuring the surface temperature in addition to the center, a rabbit was pulled out of the hat.
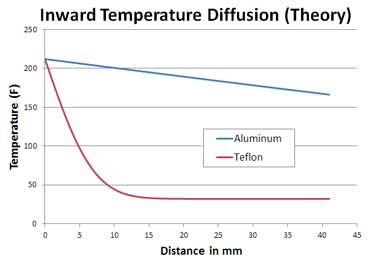
Why is the surface hotter than the interior in the first place? The reason is "thermal diffusivity", that is, how easily heat moves through the meat relative to how quickly it heats up. By analogy, imagine you are pouring water into a measuring cup. The higher the water level, the higher its "temperature". Water flows very easily, so the water level is the same from edge to edge in the cup, even during filling. Therefore, the "temperature" is the same from surface to center. And when you stop pouring, the water level barely budges. No carry-over. But now imagine you are drooling in a thick, slow to spread (e.g. "diffuse") liquid like molasses. Unlike water, the height of the molasses pool varies the farther you are from the stream. And, when you stop pouring, the molasses level magically continues to rise, as the lump in the center oozes to the edges. A treacly carry-over. So much for the analogy, but let's reinforce this argument with an experiment before hitting the grill. I placed an iron ball and a cylinder of Teflon (both around 2" in diameter) into a 325F oven. A tiny hole was drilled to their centers, providing thermocouple access. Interestingly, Teflon and meat have almost identical thermal diffusivities. And both diffuse much more slowly than a metal like iron:
(we also chose Teflon because it does not absorb water, and does not evaporatively cool like meat. More on that distinction in a few paragraphs). Heat diffusion equations reveal an expected but dramatic difference. Like water pouring into a cup, the fast diffusing iron maintains about an equal temperature profile from surface to center (blue line). But heat piles up near the surface of the slow diffusing meat/Teflon sample.
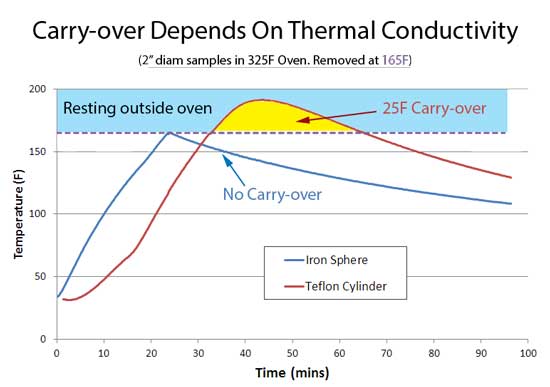
Which exactly parallels our oven experiments on lumps of iron and Teflon: Note the center temperature of the iron ball immediately climbed in the first few minutes of exposure to the oven's heat, while the Teflon ball took its time. This delay is a result of the low thermal diffusivity in Teflon, which causes heat to pile up near the surface. Fast conductivity iron, on the other hand, allowed heat to almost immediately reach the center. When the iron or Teflon samples reached 165F they were removed from the oven and allowed to cool on a rack in the kitchen. As this plot clearly demonstrates, the high thermal diffusivity iron ball not only heated up quickly, but immediately started to cool off when removed from the oven. Absolutely no carry-over at all.
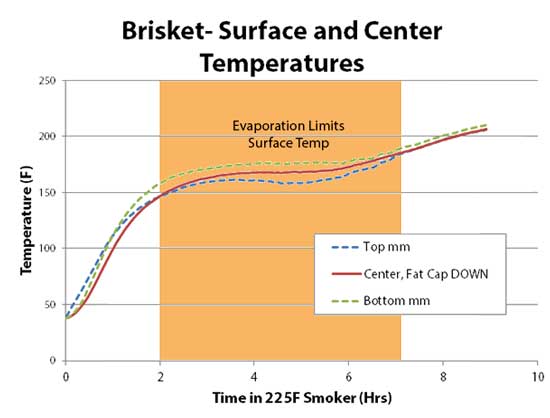
But the Teflon slug tells a different story entirely. Once removed from the oven, the center temperature of the Teflon cylinder continued to rise, carrying over by 25F until slowly entering its decline. Thus, carry-over is a phenomena limited to low thermal diffusivity materials. But food adds one more complexity- moisture. As meat cooks, moisture migrates from the interior to the exterior. Upon arriving at the meat's surface, evaporating moisture carries away enormous quantities of heat. Evaporative cooling is responsible for the stall experienced during smoking. Evaporation also slows cooking (even without stalling), and can double or triple cooking time compared to heating up dry foods. During hot air baking or smoking, evaporation prevents the meat's surface from ever1 exceeding 212F, the boiling point of water. So, while the air in an oven might exceed 212F by hundreds of degrees, food is cooked from the surface inward, and if the surface sweats and cools off, that cooler surface temperature is now the effective oven temperature. Evaporative cooling eliminates any chance of carry-over during slow and low barbecuing. It limits the maximum excursion of the surface relative to the center. In this experiment, I simultaneously measured the surface and center temperature of a beef brisket "flat" cooked in a PID controlled 225F electric smoker, fat-cap side down. Note during the first two hours of cooking, the interior temperature lagged the surface by around 20F- just as expected. But, once the meat entered the stall, the brisket surface and center "flat lined", preventing the surface from continuing to gain on the interior. Kind of like an oven sous-vide. When the evaporation rates finally declined around seven hours and the stall ended, the surface and center draw closer in temperature. In fact, carry-over is rare to non-existent at the end of a long slow cook.
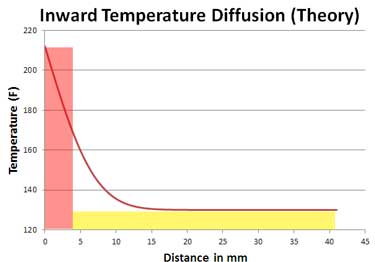
My favorite way to cook a steak, the "reverse sear", generates a uniform color from edge-to-edge. The reverse sear initially warms meat slowly and gently in a low oven, so the entire slab uniformly rises from fridge temperature to around 115F. Then, the meat is seared for just a few minutes to color and flavor the surface, allowing this extra kick of heat to carry the center through to doneness. In principle, brown on the outside, pink throughout the interior. I normally don't rest steaks after searing, preferring the taste of crisp bark, and besides, resting makes little difference in tenderness or juiciness. So I normally immediately slice the steak, cooling it slightly and locking in the medium rare color. But most people rest their meat, either intentionally or inadvertently while waiting for the side dishes to finish or for the kids to sit down at the table. It is during resting when meat will carry-over. How much carry over should we expect? A simple calculation is sufficient. Imagine you are reverse searing a two inch thick steak. Measurements and theory indicate the hot crust reaches 212F and penetrates about 1/8" after 3 minutes of searing. So instead of the complex curved temperature profile, we imagine there is a surface layer (pink bar) at 212F, and an interior (yellow bar) at the final cooking temperature of 130F. Which dramatically simplifies the analysis.
During carry over, the total amount of energy stored in the two regions evens out, taking on the average value. The calculation of the average is 8th-grade-math simple: [⅛"*212F +1 ¾ "* 130F + ⅛"*212F]/2" =140F
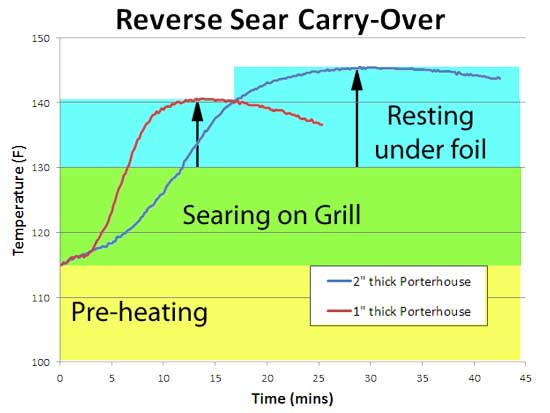
In other words, a 10F carry-over! And here are the results on actual porterhouse steaks. They were per-heated in a 225F oven until the center reached 115F. Which takes nearly an hour. Then immediately seared on a VERY hot infrared grill that reaches 1000F. The steaks were flipped once a minute2 until the center temp reached 130F- medium rare. A dark sear takes a minimum of three flips- the thinner 1" steak was flipped 3 times, the thicker 2" around 6 times, until their centers hit 130F.
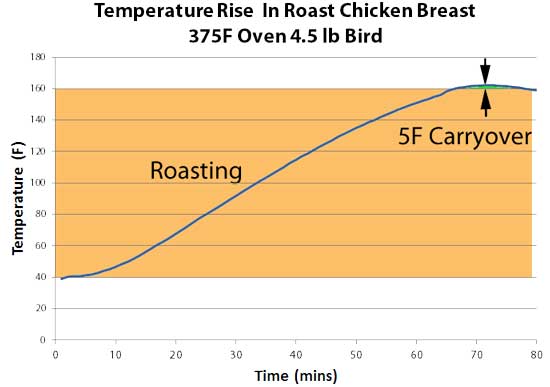
Acknowledging that most people rest their meat before slicing, each steak was rested under a foil tent. The 1" steak carried over by 10F, the 2" steak around 15F! This extra kick was enough to drive both steaks well past medium - a reluctant but necessary sacrifice in the name of kitchen science. Clearly, this much carry-over is a problem unless accounted for by the reverse-sear technique. If your target consumption temperature is 130F, and you know the carry-over for your grill is 10F or more after three flips, then pull off the grill when the interior temperature reaches 120F. Which means you might only pre-heat the steaks to 110F instead of 115F, to allow time for the three flips. The thicker steak had a larger carry-over, which is at first surprising. If the same amount of heat was deposited in the top 1/8", it would average to a lower temp in the center because there is more meat to heat up. But, in this experiment both pieces of meat started at the same pre-heated level. And to warm up a thicker piece of meat to 130F during searing doubled the time on the grill, expanding the thickness of the 212F surface layer. In the end, this caused a larger carry-over. I suggest you pre-heat thick steaks closer to their final serving temp, so the same three searing flips is sufficient to brown the surface. But, in general thicker cuts of meat result in lower carry-over. In this experiment, we roasted a whole chicken, and measured the carry-over as it rested on the counter after reaching 160F measured a half inch from the chest cavity wall. Note the carry-over is only 5F. Not too surprising- as we demonstrated in this article debunking beer-can chicken, most of the heat arrives from the surface, not from the cavity. If you employ the same 8th grade math, this time on a 2 1/2" breast heated from one side, the lower carryover emerges.
In summary:
|
||||||||||
|
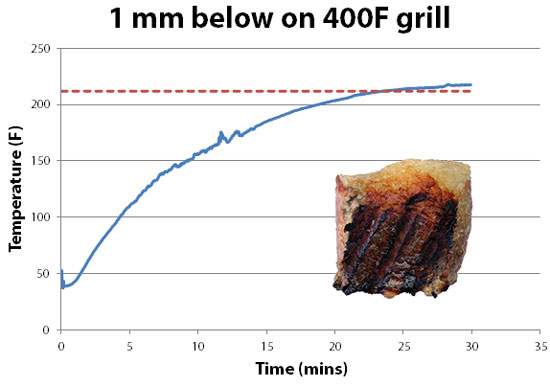
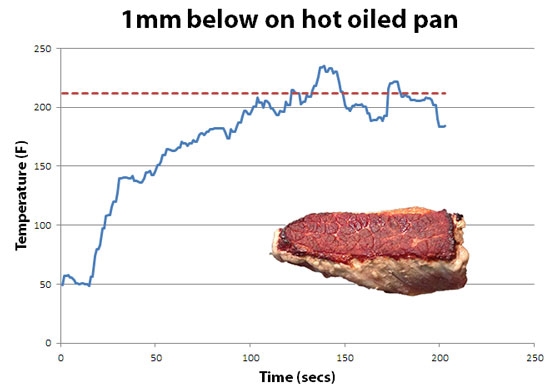
-------------------------------------------------------------------------------------------------------- 1 On a scorchingly hot griddle or grill, the surface can dry out bit and speed past boiling into charring. But not by much, so 212F is a good guide for these calculations. More specifically, on a medium temperature gas grill, the surface of a brisket slab will nicely brown, and the temperature measured a mm below the surface will rise to 220F over a few minutes:
Seared on a blisteringly hot frying pan seasoned with a bit of oil, the surface browns in seconds. The temperature record jumps around a bit as the meat spits and spurts, and can reach as high as 230F. But still averages around 212F (the dotted red line).
Rather amazing that a little bit of steam can protect a steak from contact with a 1000F surface. A testament to water's enormous latent heat of evaporation, plus a bit of Leidenfrost thrown in for good measure.
2 Why flip every minute, instead of completely searing one side, and then the other? Well, the first side will begin carrying-over while the second side is still on the grill. This means the first side's temperature begins rising a few minute ahead of the second, and is likely to end up overdone. Now, it takes a minimum amount of time on the grill for sear marks to develop and for the meat to release from the grates, so you can't flip too often or you sacrifice flavor. On a hot grill, a minute or so between flips is a perfect compromise.
|
||||||||||
 Contact Greg Blonder by email here - Modified Genuine Ideas, LLC. |
 But what about a fast cook, like searing a steak to medium rare? The traditional way to cook steak is to flop it on a hot grill. The surface is singed brown in a just a few minutes, but for any steak thicker than 3/4" or so, the slow diffusion of heat leaves the center raw. By the time the center is cooked, most of the steak is past its prime. A cross-section that runs from gray to pink.
But what about a fast cook, like searing a steak to medium rare? The traditional way to cook steak is to flop it on a hot grill. The surface is singed brown in a just a few minutes, but for any steak thicker than 3/4" or so, the slow diffusion of heat leaves the center raw. By the time the center is cooked, most of the steak is past its prime. A cross-section that runs from gray to pink.