| g e n u i n e i d e a s | ||||||
 |
 |
 |
 |
 |
 |
 |
| home | art and science |
writings | biography | food | inventions | search |
| wood fired pizza dough |
|
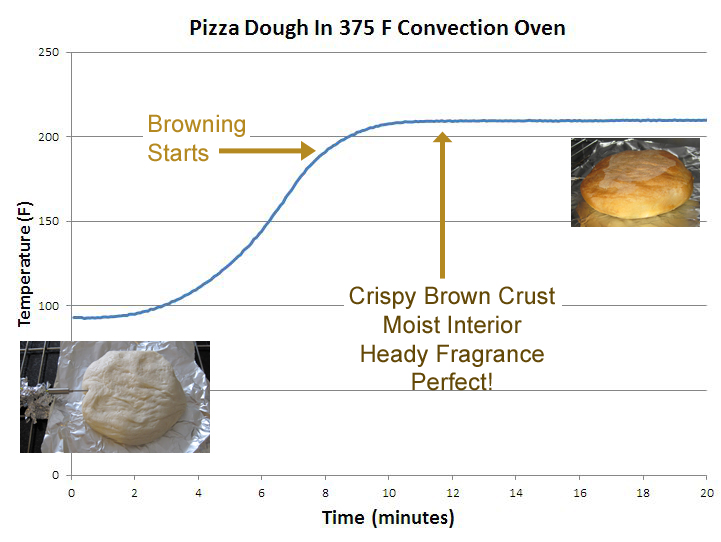
August 2011 Like barbecued meats, and even vodka infused pie crusts, pizza dough will warm up in an oven, then stall. Around the boiling point of water. But a brisket might stall at 155F. Why are these behaviors so different, and what can this tell us about baking pizza? In the case of a brisket, much of the water is "free", that is, not chemically bound to the meat and can be squeezed out of the muscle like water from a sponge. Other liquids, like myoglobin, are expelled as the proteins unravel. In either case, this copious and mobile stream continually bastes the meat, lowering its temperature below oven air by evaporative cooling. Flour dough is another beast entirely. Wet and sticky, you cannot wring "free" water from most doughs. Instead, the water is lightly bound to starch and gluten molecules in the flour, which is to say, bound to the flour about as strongly as bound to other water molecules. So, near the boiling point of water (212F) this lightly bound water boils out of the dough- the temperature is set by chemistry, rather than the mechanics of spray cooling (other water is tightly bound into new chemical compounds, but these are released at much higher temperatures). The proteins in the bread's surface also dry out and the molecules bond together, forming a crust or "bark". This dense layer retards moisture evaporation almost as well as a sheet of aluminum foil. Again forcing evaporation close to the boiling point. To see how lightly bound water affects the baking and crust development of a pizza, we kneaded a 1/3rd cup of flour, water, sugar and yeast into a standard pizza dough. After doubling in size, a thermometer was inserted, and the dough placed in a 375F convection oven. The temperature rose smoothly- by eight minutes it had reached 200F and started to brown. By 12 minutes it had reached 208F, filling the kitchen with a sweet earthy aroma-- the crust crispy brown on the outside yet moist on the inside. The perfect pizza, about to be sacrificed in the interest of kitchen science.
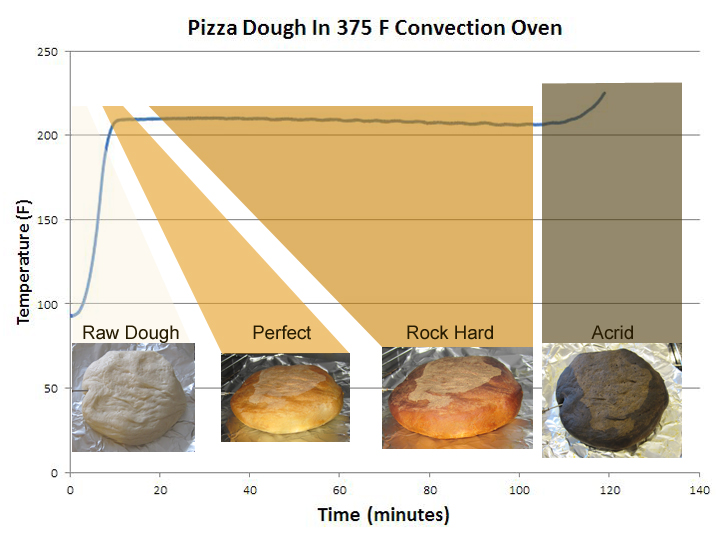
The pizza cooked and cooked for nearly another two hours. The crust gradually darkened, then grew nearly black. And the aroma shifted from sweet to acrid:
All the while stalling at 208F-amazing that a smelly hockey puck might cool itself only through evaporation for nearly two hours! (it also tells you why dough temperature is often a weak predictor of doneness). Baking in a convection ovens leaves open a window of about 5 minutes between soggy dough and an acceptable crust. But this oven runs at only at 375F- what about a wood-fired pizza oven putting out 800F? My favorite pizza style is Neapolitan- the crust is bubbly and nearly charred in spots, evenly baked on top and bottom, yet the interior still smells of warm, yeasty, almost uncooked dough. This simple pie is a miracle of timing and balance, leaping from peel to perfection in about 90 seconds. In a conventional oven, almost all the heat comes from "convection". That is, heat is transferred from the burner elements to the air, and from the air to the pizza. Convection is a relatively slow form of heating, because air is, well, "airy", and 1000 times less dense than the pizza. Simply put, the air does not transfer in much heat, because it doesn't carry much weight. In a wood-fired pizza oven, the dough is placed on the hot stones lining the oven. These bricks are at 800F or more, and the limp dough comes into intimate contact with the bricks. Heat efficiently moves by "conduction" into the dough- and very quickly, because the bricks are nearly as dense as the pizza. The same reason you sear in a frying pan but bake in an oven. But, once the pizza shrinks and bubbles, or is shifted by the pizza peel, an air gap between stone and dough appears, and this insulating layers dramatically slows heat flow. Yet the wood-fired pizza oven offers a third source of intense heat- "radiative" heat from the light emitted by the fire. Radiative heat is all around us, but usually too weak to be noticed. The bricks are emitting radiation, as is the air, and even the baker. Unlike conductive or convective heating, where energy transfer rates depend mostly on thermal conductivity, radiative heating power grows in strength according to the Stefan-Boltzmann equation, i.e. proportional to the temperature raised to the fourth power. This means a 300F (422K) oven is radiating 80x less heat intensity than an 1800F (1255K) orange wood flame (=[1255/422]4), which is why you can feel the heat from a fireplace ten feet away, but barely notice the warmth from an open oven door at the same distance. And why you must turn on the broiler in a home oven to brown dinner). It also means at some point radiative heating will outrun either conduction or convection- a little higher in temp it will burn the crust, a little lower and it will be too cool to matter. Building a proper wood fire is an art. Thus the dance of a pizza baker. If they want to produce a crust where the bottom surface (mostly heated by conduction) is exactly as crispy as the top crust (mostly heated by radiation), they have to discover the perfect location within the oven, along with the exact dough water content and bubble density, so these two heating mechanism are in sync. All the while cooking the toppings at the same time. Often the simple things in life are the most elegant.
|
|
-------------------------------------------------------------------------------------------------------- Additional articles on kitchen science can be found HERE.
|
 Contact Greg Blonder by email here - Modified Genuine Ideas, LLC. |