| g e n u i n e i d e a s | ||||||
 |
 |
 |
 |
 |
 |
 |
| home | art and science |
writings | biography | food | inventions | search |
| foiled | ||||||||||||||
|
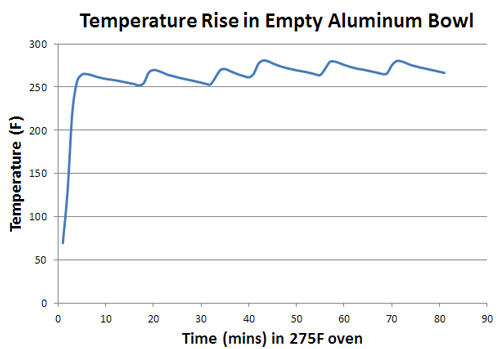
November 2011 My earliest introduction to "cooking" came buried under a campfire. At home we'd slice onions and peppers and hunks of meat, freezing them in a packet of aluminum foil in preparation for the next day's hike. Tossed under the coals after a long day outdoors, and recalled through the dusty lens of time, hunger and exhaustion, the meal rivaled any four-star restaurant. Later, I learned how to braise roasts in a dutch oven (the cast iron equivalent of a foil packet) or baste ribs swimming in a barbecue sauce foil sarcophagus. More recently (while smoking pork on an outdoor grill), I learned the value of the "Texas Crutch" to work around the "stall". Foil braising is nothing more than placing food and liquid in a sealed pouch, then indirectly cooking at a temperature greater than the boiling point of water. Reputedly, this method speeds up cooking, softens tough cuts of meat, and recycles essential flavors in the braising liquid. But what exactly is going on INSIDE the foil pack? Some people maintain the braising liquid is simmering and gently bathing its contents in a humid, fragrant fog. Others claim any water instantly bursts into steam on contact with the foil, quickly scalding the meat into submission. So let's perform a few kitchen science experiments to unravel the mystery of cooking in foil, armed with nothing more than Reynold's wrap and a thermometer. Rather than start with the complexity of a whole rib roast braising atop a mirepoix base, let's break the process down into a series of independent steps. The simplest question to ask is what happens inside an empty foil packet? In this case, the packet is a disposable aluminum foil bowl, around 8" diameter and 3" high, tightly covered with a sheet of foil. A poor man's dutch oven. Baked at 275F, the air temperature inside the foil rises up to match the oven's temperature in only five minutes.
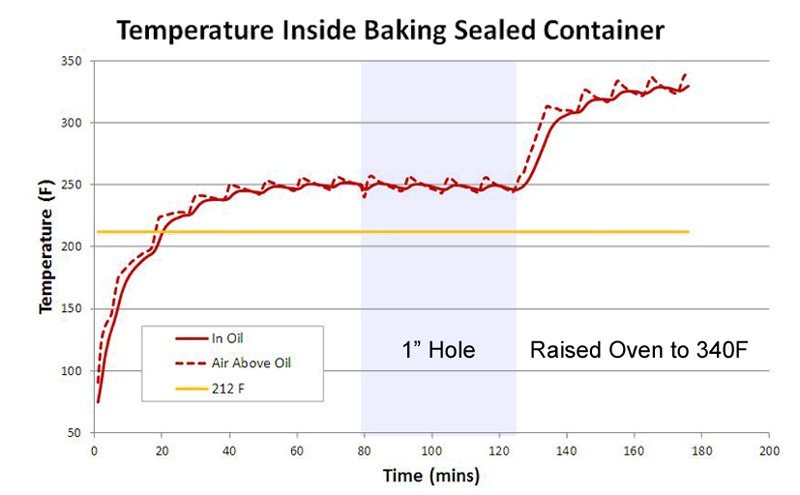
The "saw tooth" wiggles mirrors the oven's thermostat, cycling on and and off to maintain an average 275F temperature. Keep your eyes on the sawtooth- as we will discover, this periodic temperature variation provides clues to the underlying physics of braising. Now, let's add a simple food product to the foil bowl - 100 gms (3.5oz) of canola oil. With an oven set at 250F, the oil heats up a bit more slowly than an empty bowl, eventually matching the oven temp after 30 to 40 minutes. Note oil (the solid red line), cannot respond to temperature as rapidly as the lighter air (dotted red), so it tends to average out any fluctuations (the so-called "thermal mass" effect).
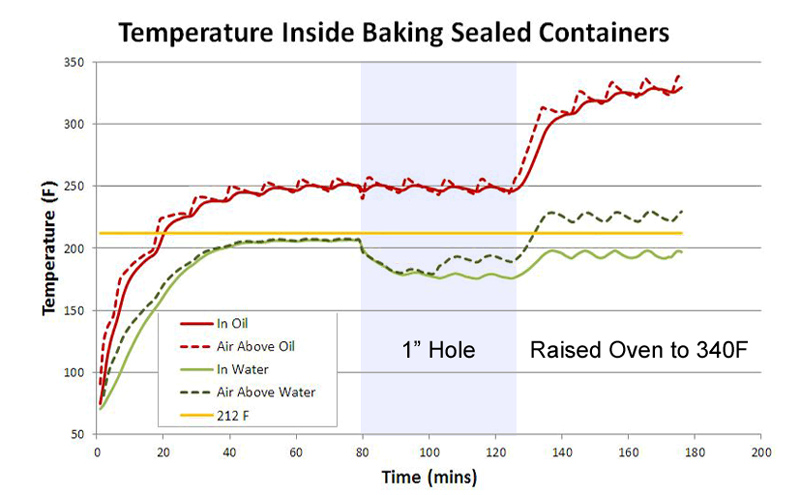
At 80 minutes I slit open a 1" hole in the foil lid, but nothing dramatic happens because the internal and external temperatures were already in equilibrium, and there is no "driving force" to create an air current through the gap. At 125 minutes I raised the oven's temperature to 340F, and after another half hour, the oil caught up with this temperature shift. Note the air temperature in the foil bowl tends to follow the oil rather than the oven temperature- that is, it takes a half hour for the air above the oil to heat, instead of 5 minutes as in the empty bowl. While aluminum is an excellent thermal conductor, air inside the bowl in direct thermal contact with the oil, and oil wins the tug of war. So far, so simple. Now repeat this experiment, replacing oil with water. Suddenly the world is more complex- and more interesting. Concentrating on the first 30 minutes, note that 100 gms of water (the green line) takes almost twice as long to heat up as 100 gms of oil (the red line). There are two reasons. Most fundamentally, water's heat capacity is twice that of oil (~4 J/gmC vs ~2 J/gmC). Heat capacity determines the temperature rise after absorbing a fixed amount of energy, and this factor of two1 explains why most chefs intuitively know a pan of oil heats faster than a pan of water.
The second reason, however, is key to understanding braising. Oil does not not evaporate, but water not only evaporates, it evaporates faster the hotter it gets, finally boiling profusely at 212F (temperature indicated by the yellow line). As water evaporates, the hottest molecules fly off after breaking free of the surface, leaving colder molecules behind, thus cooling the remaining liquid below ambient. In an open container placed in a warm oven, the water temperature rises until the increasing evaporation rate is matched by the evaporative cooling effect, and the two reach equilibrium. We call this an "equilibrium" because if the water grew any warmer, it would evaporate faster and thus cool off even more rapidly. If the water became cooler, it would evaporate less quickly, and thus warm up because fewer hotter molecules escaped. At 250 F you might expect the water bath to plateau at 170F. But, in a nearly closed container like this foil bowl, hot water vapor builds up, and eventually as many hot vapor molecules are re-entering the water as are leaving. Then the temperature plateaus closer to the boiling point. SOME water vapor still escapes around the foil edges (this is a foil packet, not a pressure-cooker), and that vapor carries away excess heat. Even so, the remaining liquid ends up simmering, not boiling, because even a small amount of water vapor carries away heat much more effectively than can be replaced by dry oven air (the "latent heat" effect). This plateau is visible above in the green curve between 40 and 80 minutes. Because of the negative feedback between evaporation rates and evaporative cooling in a closed container, the water temperature does not shadow oven temperature fluctuations. And, because any air trapped in the foil is in stronger convective thermal contact with the water than to the oven through the foil, the air too does not fluctuate. At 80 minutes I again opened a 1" hole in the foil. This gap allows hot water vapor to quickly exit the foil bowl, instead of re-entering the water layer. The water bath drops by nearly 20F due to more effective evaporative cooling. Simultaneously, a counter-current of oven air rushes into the bowl to replace the outflow of humid air, and thus the air above the water begins to shadow the oven temperature. Pretty neat. At two hours (120 minutes) I raised the oven to 340F. The moist air above the water mixed with hot oven air entering through the hole, actually exceed the boiling point. But the water bath, even in 340F oven, only rose slightly in temperature, stabilizing at 195F. From these three simple experiments, we have learned the following:
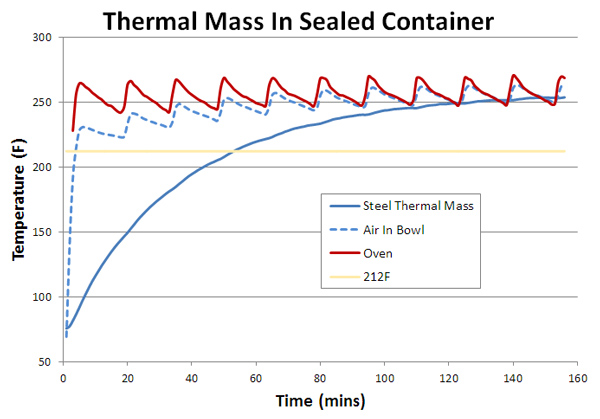
Before experimenting on actual cuts of meat, what happens if we replace water with a hunk of steel to represent just the thermal mass effect of a large portion of food? Of course, real meat also contains water, which as we now understand, behaves quite remarkably when heated. The graph below measures the temperature rise inside a foil bowl containing a 525 gms (~ one pound) hunk of steel. The red curve is the oven temperature outside the foil bowl, the blue dotted line the air temperature inside the bowl measured between the foil and the steel (the solid blue line).
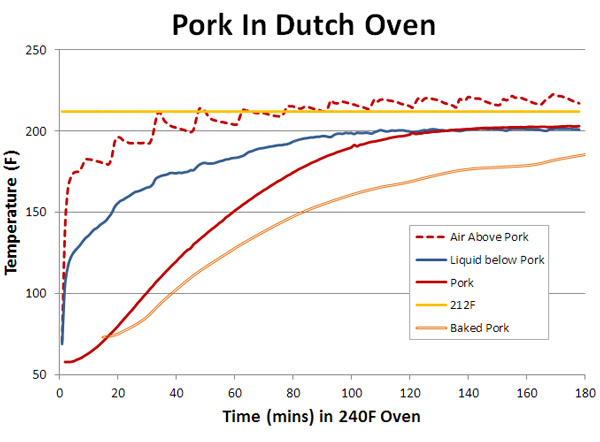
Note the air inside the foil senses an average temperature, heated on one side by the hot foil surface, and on the other side by the cooler steel mass. Because air is a very poor thermal conductor and the metal is dense and heavy, it takes over an hour for the steel hunk to attain the same temperature as the oven- you can imagine how long a 10 pound pork butt might require. Speaking of pork, we are finally ready to braise an actual piece of meat. The sample is a 225 gm(~8 oz) pork shoulder2, placed in a small dutch oven. One thermometer is inserted in the meat's center (the red curve), one placed under the meat in the braising juices (the blue curve) and the one in the air above the meat (the red dotted line). The lid is slightly ajar to permit thermocouple wires access. Other than juices released by the cooking pork, no further liquids were added. But the bottom of the pot quickly filled with a half inch layer of captured, exuded juices:
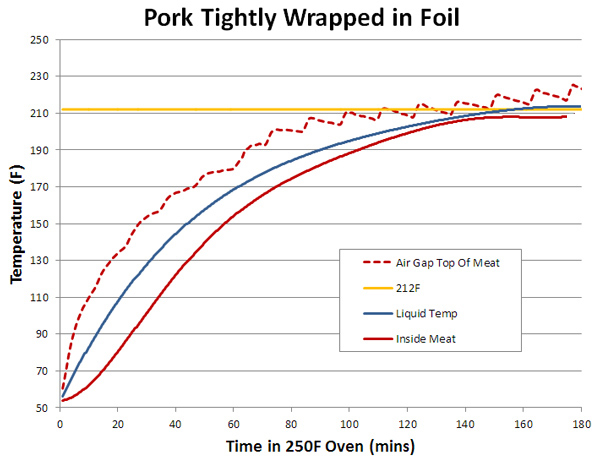
By now this behavior should be achingly familiar. For example, the liquid (blue) heats up faster than the meat (red) because it is in direct contact with the metal pot wall. The meat is heated by air (a poor conductor, 20-30x worse than water) and the liquid. So naturally it lags a bit in time. But the meat and liquid temperatures eventually plateau about 10F below the boiling point (yellow line). The air temperature also rose slightly above boiling- a 1/4" inch slit along one side of the lid provided thermocouple probe-wires access. This gap allowed warmer oven air to mix with the cooler dutch oven air. And allowed enough water vapor to exit that the liquid never reached 212F. The dutch oven also cooks more rapidly than oven baking. The brown line illustrates an identically-sized pork shoulder simply baking on an oven rack next to the dutch oven. The baked shoulder's exuded juices evaporatively cooled the meat, so it heated up more more slowly- in this case, it took about twice as long to reach the same temperature. Even though it was in a 240F oven and the braised pork in a 200F bath. But on my camping trips, and for most grill chefs, food is wrapped in foil, not braised in the bottom of a pot. How different can this be? Well, the main difference is the small (1/4") air gap between the foil and the meat, which restricts the flow of cooling water vapor, plus allowing other fluids to accumulate in their own isolated pools. Again, we test with 8 ozs of pork shoulder in a 250F oven, this time wrapped in foil:
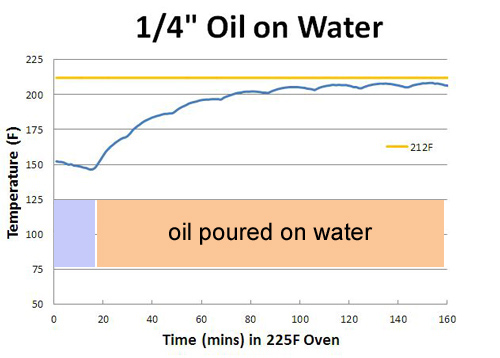
Note the liquid layer stabilized just above the boiling point, and in the air gap between the meat and the foil, climbed even higher. The reason is simple- the liquid layer contains both oils (fats) and water. These fats, as we discovered earlier, never stall around boiling because they don't evaporatively cool. So where there are pools of fat, that part of the foil envelope is significantly hotter than the water. Even water is affected by the fat, which is lighter and floats on top, acting as a localized foil cap. Such a layer creates a remarkably efficient seal- here I covered a water bowl (which was evaporatively stabilized at 150F) with a 1/4" of 150F oil. During the following hour, the water rose in temperature to 205F, with small water vapor bubbles occasionally breaking through the oil to provide cooling.
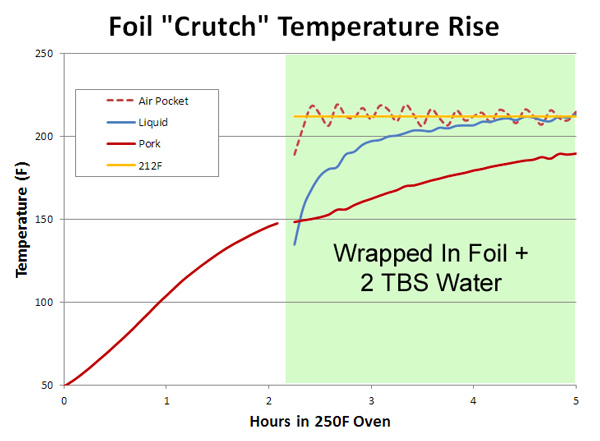
Performing the same experiment at 350F caused the water vapor to slightly heat above the boiling point, just like in a pressure cooker. These superheated water vapor bubbles burst out, splattering oil in all directions. Curiously, microbursts are why Reynold's Oven bags suggests mixing a few tablespoons of flour into the bag before cooking. The flour binds the fats to the water, preventing mini explosions that might otherwise cause the bag (which contains numerous vents) to split open. Sliced potatoes, onions and tomato paste in the bottom of a braising pot can also block evaporation. Cooking can be messy, in more ways than one. In the barbecue world, foil wrapping is often known as the "Texas Crutch". Basically, you slow cook the meat for a few hours to brown and develop smoky flavors. Perhaps until reaching 150F- actual temperatures depend on the smoker and cook's "secret" recipes. Then, wrap the meat in aluminum foil, adding in a bit of moisture (water, apple juice, sauce, ..) to prevent the 'cue from drying out, and to directly conduct heat through the foil into the bottom of the meat. Finally, after a few more hours of basting in foil until the meat is soft and tender. Either served immediately, let rest, or finished on the grill. Common wisdom holds the added moisture flashes into steam to speed up cooking. But in reality, the 150F meat's thermal mass and evaporative cooling suppresses any tendency to boil- at least for a few hours. And after a few hours, its often time to unwrap. By now you should be able to interpret this curve of a four pound pork shoulder cooked in a 250F oven on your own:
So far, we've considered only foil, but people sometimes bake chicken or apple pies in brown paper bags, and fish is often braised in parchment paper envelopes. Are these wrapping papers merely primitive substitutes for the more recent invention of aluminum foil, or will their permeability to water vapor make a significant difference? We tested four different sheets. The fry filter is used to remove solids from hot oil- the mesh is quite porous and you can easily breath through it. Parchment paper is basically paper pre-soaked in oil (today, usually food-grade silicone oil so the paper will not brown at high temperatures). It is quite dense, and is only slight permeable to moisture or oil. Then there are kraft paper bags. These bags are often stabilized with "sizing agents", sometimes clay, sometimes fish glue or plastic, so they won't easily dissolve in water. Plus, dyes and acids are used in processing, all of which are generally not food safe- be warned! Still, with a bit of lung pressure, you can gently force air through the wall of a paper bag (denser still are butcher papers- kraft paper with much higher levels of sizing agents). And finally there is our baseline impermeable, malleable aluminum foil.
We clamped a sheet of each material over a water bath, creating a sealed container. The container was inserted in a 250F oven, and the water and air temperature above the bath recorded. Here are the results for the water temperature, which reflects the internal humidity levels. The lower the water temperature, the lower the humidity:
So moisture permeability does have an effect. In a 250F oven, the more permeable the material, the more easily water evaporates from within the "sealed' container, and thus the more effectively it is evaporatively cooled. You can also see why some pit masters use butcher paper as a substitute for aluminum foil- it will eventually reach 212F, but paper is not easily punctured like aluminum foil (unfortunately, almost all butcher papers are NOT food safe at high temperatures, and the sizing agents will contribute a decidedly funky flavor. This is why paper coffee cups used to taste like socks and old cardboard). The humidity level inside a wrapped bag is lower with the more permeable films, but higher than when simply baking food on a wire rack. This means the choice of wrapping paper can make a difference, at least when cooking at low temperatures where the water bath is always below boiling. Baking a pie in a paper bag is not the same as in a dutch oven. In a later article, we'll describe how to use this effect to your advantage during smoking, but for now, concentrate on aluminum foil.... In a real, foil wrapped piece of meat:
Now that we've revealed the physics of braising, there also is the matter of taste. Which is also a matter of opinion, but here is mine:
|
||||||||||||||
|
--------------------------------------------------------------------------------------------------------
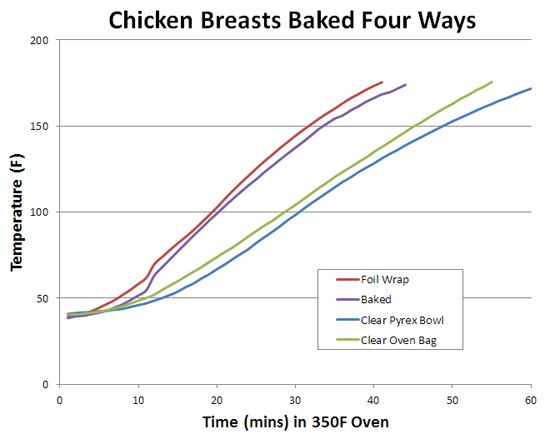
1Water is roughly the density of oil, but can store additional thermal energy in rotational vibrations of the V-shaped polar H2O molecule. This vibrational energy is not reflected in the temperature of the water, which measures the average linear kinetic energy of molecules. It's like eating candy- some of the calories go into running around (e.g. the kinetic energy of temperature), and some into fat storage (e.g. vibrational energy). 2Steel's heat capacity is ~0.5 J/gmC vs ~2.5 J/gmC for pork. So the 550 gm steel sample should increase in temperature about 2.5x as fast as 225 gms of pork (2.5/0.5*225/550). But the observed rates are similar, because the rate of temperature increase for the steel is dominated by the thermal conductivity of the air. Unlike comparing a pool of water vs. oil (both of which completely cover the bottom of the bowl, intimately connected to the oven temperature), the steel just touches down in a few places. And this limits how quickly heat penetrates the steel. 3 A weak form of browning is possible during braising. While the Maillard reaction visually begins around 325F, to some extent time can substitute for temperature. Hours of cooking at 250F will brown meat, although the mix of aromatic chemicals are not as pungent as browning at 350F. The Maillard reaction even occurs at room temperature INSIDE our bodies, working on the same amino acids and sugars that brown a Parker House roll. Additionally, braised food is often unintentionally "radiatively" heated. Normally, braising absorbs heat by conduction from air to liquid to food- so called "indirect heating". But, every surface also radiates infra-red light energy, and this light energy can be hotter than the surrounding air. Think of a cold winter day- the air temperature is below freezing, but sunlight on your cheek feels nearly as warm as in mid-July. A 350F oven is filled with radiated energy that is invisible to the eye. Much of this energy is reflected by the aluminum foil and never penetrates the packet- although a bit of radiation is emitted by the hot foil itself. So, even though its 200F inside the pot, the food "sees" a little bit of 350F browning energy from the pot's walls. The situation is more favorable when braising in a transparent oven bag or clear Pyrex dutch oven. The clear walls admit radiation, speeding up browning. Even more radiation is created a few times an hour, when the heating elements turn on bright red. This is why a turkey cooked in an oven bag achieves a light brown complexion, despite spending most of the time in a sauna. A simple experiment demonstrates this point. I placed four skin-on, bone-in 8 oz chicken breasts in a 350F oven. One breast was simply baked on a grate, one tightly wrapped in foil, one loosely contained by a clear oven bag, and one placed in a clear Pyrex bowl with a clear lid. No added water, no added flour.
They were each baked until reaching 175F, then removed from the oven. The foil wrapped chicken was white and the skin pasty, the baked chicken browned with some areas of crisp skin, and the two breasts in the clear containers managed to slightly darken, though the skin was not crisp. Another hour of baking (as with a turkey), and these two would have deepened in color:
Interestingly, the baked and foil wrapped breasts cooked at the same pace. As we discuss in a separate article on the role of humidity during baking, at high temperatures moisture cannot migrate rapidly enough to the meat's surface to evaporatively cool, so cooking time is basically unaffected.
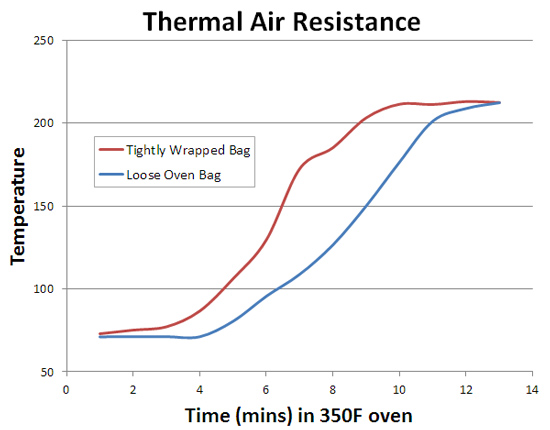
The two clear wrapped breasts cooked more slowly. This effect is not a result of some optical phenomena (the unwrapped breast is even less obstructed), but due to thermal resistance. In particular, the two breasts in clear packages were separated from the package walls by an inch or so of air (see photo above). Stagnant air has 40 times the thermal resistance of glass, and 10,000 more than aluminum. To confirm this hypothesis, I placed two identical wet sponges in clear oven bags. One sponge was tightly wrapped, the other loosely enveloped. The difference is clear:
In larger bags or with a vent, air convection reduces thermal resistance. But an inch of sealed air (just like in a thermopane window) is an effective thermal barrier. |
||||||||||||||
 Contact Greg Blonder by email here - Modified Genuine Ideas, LLC. |