| g e n u i n e i d e a s | ||||||
 |
 |
 |
 |
 |
 |
 |
| home | art and science |
writings | biography | food | inventions | search |
| sweating the details | ||
|
||
|
September 2011, May 2014 Summary- surface moisture, humidity and smoke ring growth
A classic sign of barbecue done right is the smoke ring- an intense red band, just below the bark, encircling a hunk of meat. A proxy for deep smoke flavors and slow n' low cooking. Now, it turns out smoke rings are a poor surrogate for flavor- in blind tastings, judges can't tell the difference between smoked meat with, and without a ring. It's also easy to "fake" a ring with a quick rub of sodium nitrite curing salts before cooking. So many barbecue contests have stopped granting points for smoke rings. Still, we "eat' with our eyes as well as our tongue, and smoke rings speak to tradition and aesthetics. So its worth trying to understand why, and when, smoke rings appear. Among the many hazy rumors to consider, a common belief asserts smoke rings form only below 140F. Once meat is heated above this temperature, the story goes, no additional smoke ring growth is possible. A complex "just so" story....
According to what people (mis)read in McGee, around 130F juices start flowing. And around 140F myoglobin is disrupted, losing an oxygen molecule which normally binds with iron to create meat's familiar pink color. If a nitrite bonds with unoxygenated myoglobin, it becomes the pink nitrosomyoglobin molecule and a smoke ring is formed. If it grabs onto a hydroxide molecule first, myoglobin turns into the gray hemichrome molecule. Between juices pushing nitrite compounds out of the muscle, and chemical bonding, the story of why smoke rings stop forming after 140F is complete. There are a couple of holes in this tight scenario. First, smoke ring forms on the surface, but the 140F threshold is measured in the center of the meat. Depending on cooking conditions, the thickness of the meat, and whether it stalled, 140F in the center could reflect 190F or 150F on the surface. In other words, the interior temperature is never representative of the smoke ring formation conditions. Second, myoglobin does not denature exactly at 140F. Instead, depending on a host of factors, the denaturing temperature can range from 145F to 175F. And third, the entire "surface water- NO2" theory was developed under radically different conditions than seen in most smokers, and is likely inapplicable (more details in the NO smoke ring tab). So let's get to the bottom of this assertion by experimentation. If smoke rings stop forming above 140F, then smoking partially cooked meat above 140F should halt ring development in its tracks. The test protocol is simple (if a bit tedious). Four, one pound samples of pork shoulder with different surface treatments were cooked in a pellet smoker for 12 hours at 225F, and removed when their internal temperatures reached 195F. An additional four test samples were first "pre-cooked" in a 225F electric kitchen oven, until their internal temperatures reached 155F, and their surfaces around 165F (~3 hours baking in an electric oven, of course, does not produce a ring). So well above the "magic" 140F. These pre-cooked samples were then placed into the same pellet-grill, and also allowed to smoke until reaching 195F. Four surface treatments were chosen to test the most popular theories of smoke ring development:
And here are the results:
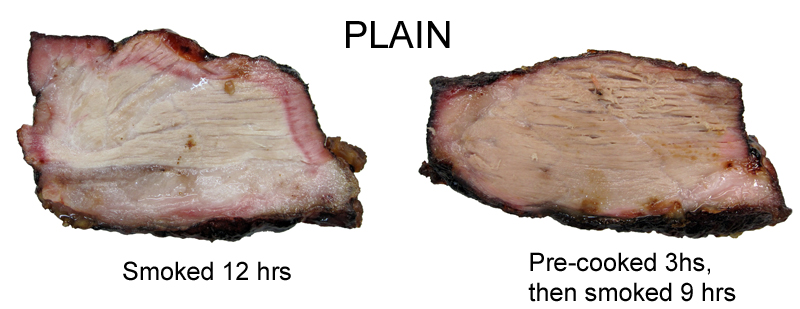
Note the strong smoke ring on the left (despite the lack of added salt from a rub), and the very faint ring on the pre-cooked sample to the right. This experiment illustrates why the theory of smoke ring development terminating after 140F has some credence. But still, there is clear evidence of a ring in places on the pre-cooked plain sample.
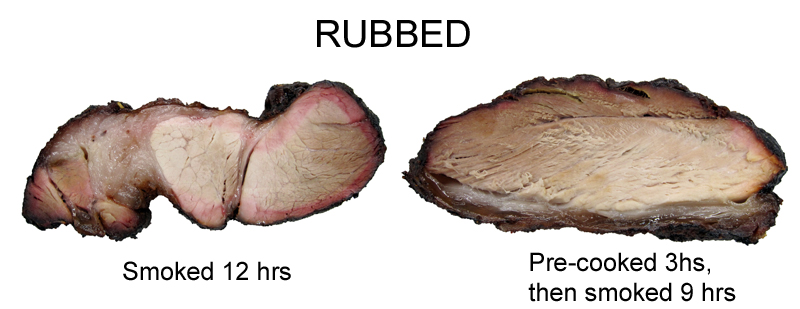
Rubbed samples, with the addition of salt and sugar, pretty much behave the same as the "plain" samples. Again, a very minor ring on the pre-cooked samples, and the paprika isn't acting like a red dye, artificially coloring the meat.
The oiled/mustard glazed sample on the left forms a smoke ring, while the pre-cooked oiled and mustard glazed sample does not form a very strong ring. But, even the pre-cooked samples show evidence of a middling ring (mostly on the sides and bottom)- contrary to the idea that ring development ends at 140F.
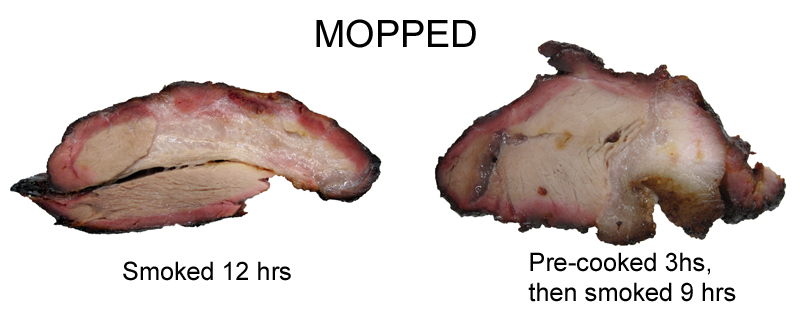
The mopped samples, whether pre-cooked or not, developed classic, intense smoke bands of uniform color. So what is going on here? In the particle flow tab above we explain how moisture encourages inward air currents, drawing smoke to the surface. Rather than flowing pointlessly around the meat. More smoke, more NO/CO and a darker ring. In fact, the edges and corners often show the ring first, because they interrupt laminar airflow and grab onto passing gases more effectively than smooth surfaces. As well as concentrate the evaporation of exuding juices. While it is true smoke ring development often stops around 140F, this observation is not the result of some weird nitric oxide chemistry or flowing juices- its due to lack of surface moisture. If the surface is dry and no longer evaporatively cooling, the interior may be at 140F while the top 1/4" is at 170F. Turning myoglobin gray before it can be stabilized by diffusing CO/NO. This is why the ring is often stronger on the bottom or sides- moisture drips to the bottom, and drying air currents are blocked by other pieces of meat or the grill gratings. Very small amounts of nitric oxide are needed to develop a smoke ring. For example, I placed a cold, raw brisket in a pellet smoker generating 30 ppm of NO for just 15 minutes. Then removed from the smoker and finished cooking in an electric kitchen oven. Not only was there a smoke ring, but a nice light smoke flavor!
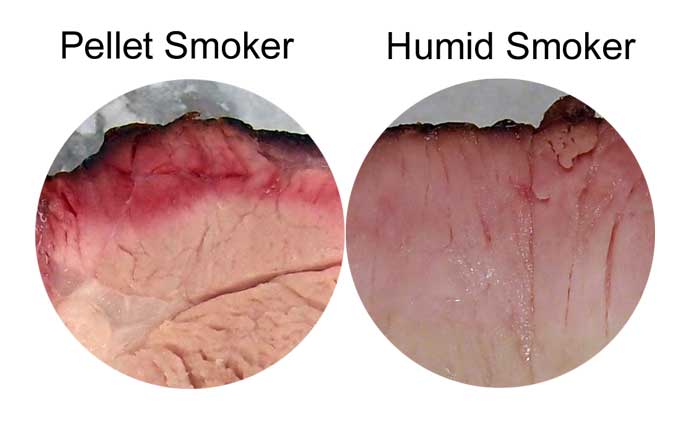
So if you can activate the surface to begin attracting smoke before the myoglobin has turned gray, the pink color is preserved. Add surface water or high humidity at almost any temperature, and the ring starts to regrow. For example, simply scoring the meat at 175F released juices, and a smoke band grew adjacent to the cut edges. You can observe this effect very clearly by comparing the shape of a pork tenderloin smoke ring generated in a pellet cooker (which tends to quickly dry out the meat's surface due to the rapid air-flow rates) and a specially modified smoker with very high humidity levels. Note the high humidity ring never stopped growing, and is nearly 3x deeper than the pellet smoker ring after 8 hours. It's also darkest near the surface, as you would expect if smoke chemicals are continually absorbed by the meat, and then diffused inward. So people who claim it doesn't matter if you stop smoking after 3 hours are right, unless you maintain high humidity levels..
Myoglobin itself, depending on meat species and treatment, will not denature until 170F. Any intact myoglobin exposed to NO/CO turns pink, as long as the surface temperature is below the denaturing point. So there is opportunity for the ring to continue to grow. Providing the surface is moist enough to attract and hold onto NO/CO. The keys to producing a strong smoke ring are based on retaining a moist surface, through a combination of:
|
||
|
|
||
Contact Greg Blonder by email here - Modified Genuine Ideas, LLC. |